J.ophthalmol.(Ukraine).2018;4:49-55.
|
https://doi.org/10.31288/oftalmolzh201844955 Received: 18 May 2018; Published on-line: 31 August 2018 Structure of the chorioretinal complex in the rabbit eye after vitrectomy. Report 2. Vitreous cavity irrigation with different temperature solutions for 60 minutes O.S. Zadorozhnyy1, Cand Sc (Med), R.E. Nazaretian1, V.V. Myrnenko1, V.A. Naumenko1, Dr Sc (Med), Prof., E.V. Maltsev1, Dr Sc (Med), Prof., N.V. Pasyechnikova1, Dr Sc (Med), Prof., V.V. Shafranskii2, Dr Sc (Med), Ass. Prof. Received: 18 May 2018 Published: 31 August 2018 1 Filatov Institute of Eye Diseases and Tissue Therapy, NAMS of Ukraine; Odessa (Ukraine) 2 Bohomolets National Medical University; Kiev (Ukraine) E-mail: laserfilatova@gmail.com TO CITE THIS ARTICLE: Zadorozhnyy OS, Nazaretian RE, Myrnenko VV, Naumenko VA, Maltsev EV, Pasyechnikova NV, Shafranskii VV. Structure of the chorioretinal complex in the rabbit eye after vitrectomy. Report 2. Vitreous cavity irrigation with different temperature solutions for 60 minutes. J.ophthalmol.(Ukraine).2018;4:49-55. https://doi.org/10.31288/oftalmolzh201844955
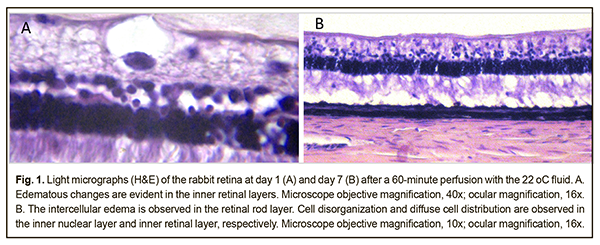
Background: It remains poorly understood what should be the temperature of the irrigation solution for intraocular surgery and how long it is reasonable to use irrigation solutions during vitrectomy. Purpose: To investigate the structure of rabbit chorioretinal complex after vitrectomy with the use of irrigation solutions having different temperatures for 60 minutes. Materials and Methods: Twelve Chinchilla rabbits (24 eyes) were divided into two experimental groups and one control group. The two experimental groups, each of 5 rabbits (10 eyes), underwent a three-port pars plana vitrectomy with either 22°C or 5°C irrigation solution. Two intact rabbits (4 eyes) were used as controls for comparison. The duration of irrigation/aspiration was 60 minutes. Specimens for histology were obtained at days 1 and 7 after surgery. The chorioretinal complex (i.e., the choroid and retina) was histologically examined. Results: After vitrectomy with 60-minute continuous vitreous cavity cooling with either the 22°C or 5°C irrigation solution, we found retinal structural changes in the form of uneven edema, (a) predominantly in the inner retinal layers, at day 1, and (b) in both the inner and outer retinal layers, at day 7. There were destructive changes in the inner retinal layers on some slides from rabbits of both groups. In addition, the choroid appeared somewhat thickened. Conclusion: In the rabbit eyes after vitrectomy with 60-minute continuous vitreous cavity cooling with either the 22°C or 5°C irrigation solution, we found the uneven retinal edema that is likely to be mediated by aseptic inflammatory response to cooling-induced and mechanical damage to the retina, and might be also associated with the composition of the irrigation fluid. Keywords: vitrectomy, intraocular temperature, rabbit eye, chorioretinal complex Introduction Pars plana vitrectomy has been the gold standard for the treatment of vitreoretinal disorders [1]. In spite of advances in surgical technologies, there are still a number of issues to be solved with regard to increased risk of intra- and post-operative complications. Thus, phototoxic damage and even thermal damage to the retina during vitrectomy has been reported. This is not surprising, since during vitrectomy procedures, the media provide no protection of the retina from the light of the endoilluminator [2]. In addition, toxic effects of dyes used during vitrectomy on the retinal neuroepithelium [3]. Moreover, irrigating solutions of various compositions have been reported to negatively affect electrophysiological and structural retinal characteristics after vitrectomy [4], and mechanical damage to the inner retina from air infusion during vitrectomy have been described [5]. Intraocular pressure rise and systemic arterial pressure decrease during vitreoretinal surgery can cause a dangerous decrease in perfusion pressure resulting in intraoperative ischemic retinal and optic nerve damage [6]. Therapeutic controlled hypothermia has been successfully applied in various medical fields (like cardiac surgery, neurosurgery, resuscitation science, and neonatology) for improving brain cell resistance to ischemic conditions [7-10]. Ophthalmological surgical procedures are performed under conditions of uncontrolled artificial local ocular hypothermia since the temperature of the intraocular media approaches that of the body, and the temperature of irrigation solutions used during vitrectomy is commonly lower than that of the intraocular media. Usually, the temperature of the irrigating solution corresponds to that of the operating room environment and is not controlled during surgery [11]. The use of a room-temperature (22-24 °С) infusion solution in vitreoretinal surgery results in a drop of intraocular temperature to deep hypothermia levels (below 30 °С) [8, 12]. The literature is, however, scant on safety assessment for different temperature regimens in eye surgery [13]. It remains poorly understood what should be the temperature of the irrigation solution for intraocular surgery and how long it is reasonable to use irrigation solutions during vitrectomy. Understanding the dynamics of structural changes in chorioretinal structures under conditions of hypothermia will enable (1) the development of a controlled ocular hypothermia technology, (2) more effective use of benefits of low temperatures in eye disease treatment, and (3) reductions in the rates of some intra- and post-operative complications. The purpose of the study was to investigate the structure of rabbit chorioretinal complex after vitrectomy with the use of irrigation solutions having different temperatures for 60 minutes. Materials and Methods Twelve Chinchilla rabbits (24 eyes; weight, 2.5-3.5 kg) were included in this study and divided into two experimental groups and one control group. The two experimental groups, each of 5 rabbits (10 eyes), were subjected to vitrectomy with either 22°C or 5°C irrigation solution. The ambient operating room temperature was between 22 °С and 24 °С. Two intact rabbits (4 eyes) were used as controls for comparison. A 23-G three-port pars plana vitrectomy was performed using the Alcon Accurus 400VS vitrectomy system (Alcon Laboratories, Fort Worth, TX). Technique The surgical site was prepared with antiseptic solution and epibulbar anesthetic was administered. Thereafter, core and peripheral vitrectomy was performed with cutting rates of 1500-1800 cuts/min, aspiration pressure of 150 mm Hg, and irrigation pressure of 20 mm Hg. The duration of irrigation/aspiration was 60 minutes. Balanced Ringer’s lactate was used as an intraocular irrigating solution. Cold (5 °С) fluid was prepared by cooling the solution inside the irrigating tube with gel packs that were located outside the tube, and thus cooling was performed in close proximity to the surgical site. Room-temperature (22 °C) fluid was obtained by placing the bottles with solution in the operating room for several hours before surgery. The temperature of the irrigating solution delivered into the eye was monitored and controlled during surgery. A thermoelectric device [14, 15] developed by the Institute of Thermoelectricity of the NAS of Ukraine and MES of Ukraine, and the Filatov Institute was used for measuring temperatures of various ocular structures, irrigating solution, and operating room. In addition, a smartphone attached infrared thermography system, FLIR ONE, (FLIR Systems, Wilsonville, OR) was used to monitor and control the temperature of the irrigating solution. After a lid speculum was placed, epibulbar anesthetic was administered, the three ports were created, and a temperature measuring probe was introduced into the vitreous cavity through a standard 23-G pars plana port. Temperatures in various structures of the ciliary body were measured before surgery and at different surgery time points. In addition, rectal temperature measurements were taken, and air temperature and relative humidity in the operating room were registered. The experimental temperature data was subjected to statistical analysis. Data is presented as mean ± standard deviation (SD). Statistical analyses were conducted using Statistica 10.0 (StatSoft, Tulsa, OK, USA) software. The level of significance p ≤ 0.05 was assumed. All animal experiments were performed in compliance with the Law of Ukraine on Protection of Animals from Cruel Treatment No. 3447-IV dated 21.02.2006 and European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes from the European Treaty Series (Strasbourg, 1986), and approved by a local Bioethics Committee of the Filatov Institute. The animals were housed and bred conventionally. Each animal underwent biomicroscopy and ophthalmoscopy at baseline, immediately after surgery, and then daily until euthanasia. Prior to surgery, animals were anesthetized with thiopental sodium 10% (1.0 mL/kg, intramuscularly). Immediately thereafter, both eyes received a drop of proxymetacaine HCl (0.5%) for topical anesthesia. The pupils were dilated with atropine sulphate. After surgery, a drop of sulfacyl natrium 20% and a drop of Ofloxacin 0.3% were applied to each eye four times daily during the observation period of 1 to 7 days. Histology was performed at the Pathology and Electronic Microscopy Laboratory of the Filatov Institute. Three animals from each experimental group of five rabbits were euthanized on the first postoperative day, and the remaining two rabbits in each experimental group were euthanized on the seventh postoperative day. Immediately after euthanasia of each rabbit, eye globes were enucleated and fixed for 24-48 hours in 10% neutral formalin. This was followed by processing, embedding in paraffin, sectioning to 5 μm, mounting and hematoxylin and eosin staining in a routine manner. The chorioretinal complex (the retina and choroid) was histologically examined by light microscopy. Results At baseline, mean rectal temperature was 38.1 ± 0.5 °С in the 22°C group and 38.11 ± 0.3 °С in the 5°C group (p = 0.97). In the 22°C group and the 5°C group, mean midvitreous temperature was 37.39±0.6 °С and 37.3±1.2 °С, respectively (p = 0.89), at baseline, and decreased to 25.95±0.6 and 10.8±0.8 °С, respectively, (p < 0.0001) at minute 10 of vitreous cavity irrigation during surgery. Therefore, compared to baseline, midvitreous temperature decreased by 11.4 °С and 26.5 °С (p < 0.0001), respectively. Mean vitrectomy time was 4 minutes, and mean irrigation/aspiration time was 60 minutes. In both groups, midvitreous temperature was not significantly different from baseline at 10 minutes after completion of irrigation. No corneal changes were observed, and lens clarity was maintained in all eyes during surgery. In all operated rabbit eyes, pallor of the retinal site in the projection of infusion fluid flow was ophthalmoscopically visible during the irrigation stage. These retinal changes, however, regressed the day after surgery. At day 1, histological examination found no substantial changes in the posterior eye tissues (including the medullary ray region) on the slides from the animals of the 22°C group. In addition, although no marked edema was noticed in the retinal layers (including the peripheral retina), histological signs of edema were observed at some inner retinal locations (in particular, in the ganglion cell layer (GCL)) on the slides (Fig. 1 A). Moreover, the choroid appeared somewhat thickened.
At day 7, signs of edema were common in the photoreceptor layer (PHL, Fig. 1B), whereas edematous cavities at some inner retinal locations (e.g., in the inner nuclear layer (INL) and GCL) were uncommon on the slides from the animals of the 22°C group. In addition, normal neuronal arrangement in the retinal INL was common, whereas disorganization of the cells of this layer with increased cell-to-cell distances was occasional (Fig. 1B).
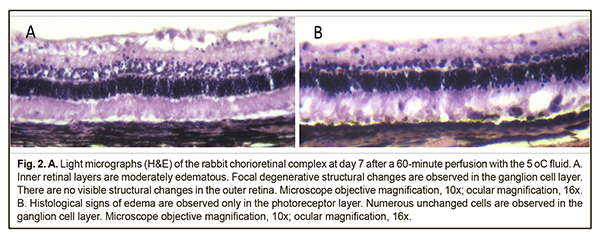
At day 1, histological examination found marked edema in the inner retinal layers (from the GCL to the INL) and mild edema in the PHL on the slides from the animals of the 5°C group. In addition, there were retinal sites with less severe edema both in the PHL and inner retinal layers. It should be noted that the structure of the retina underlying a rather thick medullary ray (the ray is 2-2.5 times as thick as the underlying retina) was practically normal. At the same time, not only edema, but also other retinal structure abnormalities were absent in the rabbit’s visual streak that contains abundant ganglion cells and is considered as an analogue of the human macula. In addition, the choroid appeared somewhat thickened. At day 7, histological examination found (a) practically no changes in the choroid and sclera, and (b) mild edema in the inner retinal layers (the GCL and the retinal nerve fiber layer, RNFL) on the slides from the animals of the 5°C group. In addition, on some slides, there were destructive changes in the inner retinal layers in the absence of edema in the PHL (Fig. 2A). Moreover, although the structure of the retina underlying a thick vascularized visual streak was still practically normal, there were mild signs of edema in the PHL, but not in the GCL, in some other retinal sites (Fig. 2B). Discussion At day 1 after 60-minute cooling of the vitreous cavity with irrigation solution, retinal edema was commonly mild on the slides from the animals of the 22°C group, and, consequently, all the structures of the chorioretinal complex appeared not substantially different from normal. Edematous changes were occasional in retinal locations, predominantly in the inner retinal layers. The neurons of the GCL (i.e., the neurons adjacent to cooling fluid flow) mostly appeared normal. However, at day 7, the signs of retinal damage on the slides from the animals of this group were more marked than before. In addition, retinal edema was more marked in the PHL, and was occasional in the inner retinal layers. Moreover, in some retinal locations, nucleus-to-nucleus distances of nearest-neighbor neurons from the INL were increased, which is likely another sign of edema in this layer. There were, however, patches of the retina with preserved structure. It is noteworthy that, at day 1, there were more apparent histological signs of edema in the internal retinal layers than in the PHL on the slides from the animals of the 5°C group. The finding of preserved structural integrity of the retina underlying the medullary ray is well understood as opposed to the reason for the absence of not only edema but also other signs of structural damage to some other retinal sites (e.g., even the visual streak). By day 7, the retinal edema was histologically evident, but was unevenly distributed, both among retinal layers and along the retina (Fig. 1). The edema was most prominent in the GCL and PHL, and a higher degree of edema was more common in the internal retinal layers than in the PHL. We hypothesize that retinal edema in the rabbit after vitreoretinal surgery was mediated by aseptic inflammatory response to cooling-induced and mechanical damage to the retina. One cannot say that the amount or location of edema substantially changed on day 7 as compared to day 1. In addition, no signs of the degenerative neuronal changes characteristic for some retinal disorders like diabetic retinopathy were found. As it was evident that by day 7 the retinal inflammatory process did not subside, the question arises: when will the inflammation subside or become chronic? Answering this question requires further experimental research. The destructive changes that were occasionally found in the inner retinal layers on the slides from both groups of animals were likely to be associated with mechanical retinal damage from a jet of infusion fluid. Thus, in the operated rabbit eyes of either the 22°C or 5°C group, pallor of the retinal site in the projection of infusion fluid flow was ophthalmoscopically visible during the irrigation stage. The mechanical internal retinal damage by air infusion during vitrectomy has been reported in rabbit eyes at the opposite side of the infusion cannula [5], and the authors noted that increased infused air pressure was accompanied by a significant increase in the area of retinal damage. Another important finding of our study was that the edema-related changes were unevenly distributed along the retina, from the optic disc to the ora serrata (i.e., retinal patches with more or less apparent signs of edema were found). The reason for such a difference remains unclear except for the retina underlying medullary rays. In our opinion, nasal and temporal medullary rays protect the underlying retina from both cooling-induced and mechanical damage, being approximately at least twice as thick as the underlying retina and well vascularized. It has been reported that, in rabbit eyes, vitrectomy with three hours of hypothermic perfusion with 2°C lactated Ringer's solution resulted in retinal damage [13]. In addition to the above, it should be taken into account that a portion of irrigation fluid introduced into the eye is the coolest at the point of infusion, and its temperature may somewhat increase as the portion goes away from this point. It, however, appears unlikely that these fluctuations in irrigation fluid temperature could be substantial enough to influence the degree of changes found at different retinal sites, especially given that retinal edema was observed on the slides from both the 22°С and 5°С groups of rabbits. As for the choroid, it was found somewhat thickened, especially on the slides from the 5°С group, which could also represent aseptic inflammation. Certainly, not all structural changes in the retina could be explained just by cooling-induced or mechanical damage. One cannot exclude that various compositions of irrigation solutions can contribute to damage to the chorioretinal complex. A number of authors [16] have noted that light and electron microscopy provided no evidence of marked structural changes in the retina after vitrectomy with short-term vitreous cavity irrigation with balanced salt solution. Since the report [16] does not specify the temperature of irrigation solution, it is likely that room temperature solution was used. In our previous report [17], we have found that in rabbit eyes subjected to a thirty-minute vitreous cavity cooling with the 22 °C or 5°С irrigation solution, there was no light microscopy evidence of structural changes in the retinal and choroidal components compared to intact animals. In their recent paper [4], Dayem et al compared the effect of three different intraocular infusion solutions (room-temperature Lactated Ringers (LR), balanced salt solution (BSS) and balanced salt solution-plus (BSS-Plus)) used during vitrectomy on postoperative retinal histology of 11 rabbit eyes. The first of the relevant key features of their technique that can be emphasized is the use of the 20-G armamentarium. In addition, the infusion was not continuous (throughout the 60-minute total vitrectomy time, each 5-minute infusion period was followed by a 15-minute non-infusion period, and vice versa), and biopsies were taken from the retina of vitrectomised rabbit eyes 15 weeks postoperatively. In the LR eyes, 2 had normal retinas and 5 of 7 had histological changes in the inner and outer retina that included vacuolization of either the inner or outer retina or both up to complete disruption. Each of the two retinas from BSS-irrigated and BSS-Plus-irrigated eyes demonstrated no gross histological abnormalities [4]. In our current study, disorganization of the cells of the INL and edema in the PHL were observed in both the 22°C and 5°C groups. Therefore, the development of controlled ocular hypothermia technology for vitreoretinal surgery requires further research aimed at identification of the time and temperature regimes for the most safe and effective cooling of the intraocular structures. Conclusion First, after vitrectomy with 60-minute continuous vitreous cavity cooling with either the 22°C or 5°C irrigation solution, we found retinal structural changes in the form of uneven edema (a) predominantly in the inner retinal layers, at day 1, and (b) in both the inner and outer retinal layers at day 7. Second, retinal edema in the rabbit after vitrectomy with 60-minute continuous vitreous cavity cooling with either the 22°C or 5°C irrigation solution is likely to be mediated by aseptic inflammatory response to cooling-induced and mechanical damage to the retina, and might be also associated with the composition of the irrigation fluid.
Finally, duration of vitreous cavity irrigation should be controlled during vitrectomy in order to decrease the risk of postoperative complications relevant to the chorioretinal structures. References
|